Let’s start with a few sentences taken from Wikipedia:
“Radiocarbon dating is a method for determining the age of an object containing organic material by using the properties of radiocarbon, a radioactive isotope of carbon (carbon-14, or 14C). […]
In nature, carbon exists as three isotopes: two stable, non-radioactive (carbon-12 or 12C), and carbon-13, or 13C), and one radioactive (carbon-14, or 14C, also known as “radiocarbon”). The half-life of 14C (the time it takes for half of a given amount of 14C to decay) is about 5730 years, so its concentration in the atmosphere might be expected to decrease over thousands of years, but 14C is constantly being produced in the upper atmosphere, primarily by galactic cosmic rays. […]
During its life, a plant or animal is in equilibrium with its surroundings by exchanging carbon either with the atmosphere or through its diet. It will, therefore, have the same proportion of 14C as the atmosphere, or in the case of marine animals or plants, with the ocean. Once it dies, it ceases to acquire 14C, but the 14C within its biological material at that time will continue to decay, and so the ratio of 14C to 12C in its remains will gradually decrease. Because 14C decays at a known rate (-50% after 5730 years), the proportion of radiocarbon can be used to determine how long it has been since a given sample stopped exchanging carbon: the older the sample, the less 14C will be left.”
Let’s not go here into the details of how 14C isotopes are counted with accelerator mass spectrometry, to find the ratio 14C/12C that will lead us to the age of the sample. Let’s just remember that the initial ratio, when the sample was still alive, was around 10-12 and that we are looking for a fraction of that. This obviously requires some quite sophisticated equipment.
This would be nice and clear if the level of 14C in the atmosphere had remained constant over time. In fact, the level of 14C in the atmosphere varied significantly and as a result, the values provided by the dating above must be corrected by calibration curves that convert a measured 14C age into an estimated calendar age. This calibration is based on dendrochronology which is a surprisingly accurate method for dating tree-samples up to 14 000 years old (in 2020). The latest calibration curve was published by Paula Reimer (2020) with a large group of researchers from all over the world.
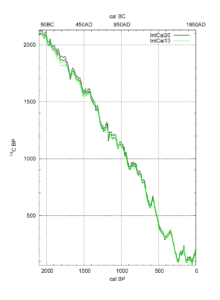
(Reimer, P., et al., 2020).
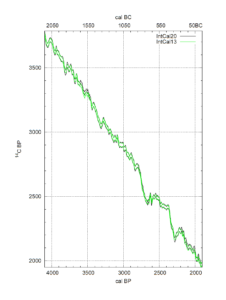
(Reimer, P., et al., 2020).
Let’s have a look at the resulting calibration curves (‘IntCal20’) for the periods 0-2000 BP and 2000-4000 BP (Before Present, i.e., before 1950 AD). The horizontal axes show “cal BP” (below) and cal BC (top) with 0 cal BP = 1950 AD on the right side. The vertical axis shows “14C BP” which is the dating resulting from the spectrometer measurement.
Suppose you found 1000 14C BP: that will convert to ca. 900 cal BP, or ca. 1050 cal AD.
Suppose you found 2500 14C BP: that will convert to ca. 2700 to 2500 cal BP, or ca. 750 to 550 cal BC. In this case, the calibration curve yields an unprecise 200-year span.
Suppose you found 2950 14C BP: that will convert to ca. 3150 to 3050 cal BP, or ca. 1200 to 1100 cal BC. This is around the end of the Late Bronze Age (LBA), and again, we have an unprecise conversion of the 14C date to the calendar date. This is unfortunate as debate on the dating of Sea Peoples’ invasions during several decades near the end of LBA is still ongoing and one cannot yet make a better guess than a range of 1250 to 1100 BC (Manning in Fischer & Bürge, 2017).
These examples show that radiocarbon dating is not a straightforward exercise and the least we can say is that it is far from being “high resolution” in time, as this would require an annual precision or better (even decenial precision cannot be provided by radiocarbon dating).
Let’s now turn to marine conditions that require a specific calibration curve (called ‘Marine20’) because of the so-called ‘reservoir effect’. Indeed, 14C contained in atmospheric CO2 transfers to the ocean by dissolving in the surface water as carbonate and bicarbonate ions; at the same time carbonate ions in the water are returning to the air as CO2. This exchange process bringing 14C from the atmosphere into the surface waters of the ocean takes time, and even more time is needed for deeper water layers. This introduces a bias in the radiocarbon dating method because what you measure is ‘old carbon’ that is older than the terrestrial carbon found on nearby land.
The surface-waters calibration curve (‘Marine20’) was thus prepared from a comparison of the dating of marine samples (corals) with that of nearby terrestrial samples.
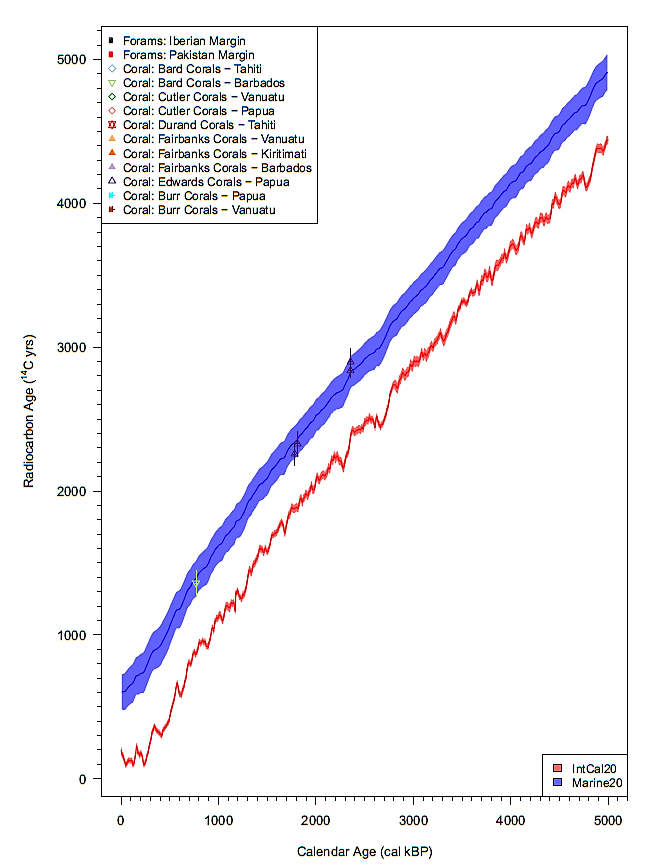
It shows a (minus) ca. 550 ± 100-year shift for the Holocene era (Heaton, et al., 2020). This curve is a global average of the marine reservoir age (also called ‘MRA’, or ‘R’ or ‘R-age’), and it can be understood that because of local atmospheric and oceanic current patterns, specific locations will deviate from this average. For this reason, a correction to the global averaged Marine20 calibration curve is used in the form of a ‘Delta-R correction’. This work is still ongoing and is collected on an online database (http://calib.org/marine/index.html ). A quick look at this database for the Mediterranean area, shows that most corrections range from +100 to -250 years, emphasising that for the Holocene era in shallow water marine conditions (< 75 m deep), radiocarbon dating yields a quite limited precision of at best ± one century.
Two (quite user-unfriendly!) softwares are available for this calibration procedure: http://calib.org/calib/calib.html and https://c14.arch.ox.ac.uk/oxcal/OxCal.html.
Further reading:
FISCHER, P. & BÜRGE, T., 2017, “Sea Peoples Up-to-Date, New Research on Transformations in the Eastern Mediterranean in the 13th–11th Centuries BCE”, Proceedings of the ESF-Workshop held at the Austrian Academy of Sciences, Vienna, 3–4 November 2014, (406 p).
GOIRAN, J-P., et al., 2021, “Developments in Geoarchaeological Research, Methodologies and Applications in Harbour Maritime Archaeology, in “Seasides of Byzantium, Harbours and Anchorages of a Mediterranean Empire”, Byzanz zwischen Orient und Okzident 21 (Mainz 2021), (p 109-129).
HEATON, T., et al., 2020, “Marine20-The marine radiocarbon age calibration curve (0-55,000 cal BP)”, Radiocarbon, Vol 62, Nr 4, 2020, (p 779–820).
KANIEWSKI, D., et al., 2019, “300-year drought frames Late Bronze Age to Early Iron Age transition in the Near East: new palaeoecological data from Cyprus and Syria”, Regional Environmental Change, 19, (p 2287-2297).
MANNING, S., et al., 2017, “Dating the End of the Late Bronze Age with Radiocarbon: Some Observations, Concerns, and Revisiting the Dating of Late Cypriot IIC to IIIA”, in “Sea Peoples Up-to-Date”, Fischer & Bürge, edt, (p 95-110).
REIMER, P., et al., 2020, “The IntCal20 Northern Hemisphere radiocarbon calibration curve (0–55 cal kBP).”, Radiocarbon, Vol 62, Nr 4, 2020, (p 725–757).